AskGene IGCC 2023 Oral Presentation: Latest Results of ASKB589 Phase I/II Trial
Camarillo, California, June 16, 2023 – AskGene Pharma Inc. today gave an oral presentation at the 15th International Gastric Cancer Congress (IGCC 2023) in Yokohama, Japan of the latest clinical results from NCT04632108 for ASKB589, an anti-CLDN18.2 antibody, for the treatment of solid tumors.
Presentation Details
- Title: A Phase I/II Study of ASKB589 (Anti-CLDN18.2 Monoclonal Antibody) in Patients with Solid Tumors
- Session: Oral 26 – Clinical Study
- Date and Time: Friday, June 16, 2023 16:50-17:22 Local Time
- Abstract Number: 01074
- Leading PI: Dr. Lin Shen, Professor from Peking University Cancer Hospital
NCT04632108 is a first-in-human study evaluating the safety, tolerability, pharmacokinetics, immunogenicity and preliminary efficacy of ASKB589 as a monotherapy and in combination with chemotherapy in patients with advanced solid tumors. The study includes ASKB589 monotherapy dose escalation and expansion (Part A) and dose escalation and expansion of ASKB589 combined with CAPOX (capecitabine and oxaliplatin) (Part B). Patients in dose escalation were enrolled regardless of the expression of CLDN18.2, while patients in the dose expansion were only enrolled if their tumors tested positive for CLDN18.2 by a diagnostic assay at a central laboratory.
As of April 25, 2023, for the 24 patients with measurable lesions, at least one post-treatment tumor assessment, medium to high CLDN18.2 expression, and treatment with 6 and 10 mg/kg ASKB589 in combination with CAPOX (capecitabine and oxaliplatin ), the investigator-confirmed objective response rate (cORR) was 79.2%, and the disease control rate (DCR) was 95.8%.
Efficacy of 1st Line Treatment with ≥ 6mg/kg ASKB589 and CAPOX for CLDN18.2 Positive G&GEJ Cancer Patients (n=24)
| Dataset with ≥ 1 Efficacy Evaluation | 6mg/kg (N=13) | 10mg/kg (N=11) | Total (N=24) |
| Objective Response Rate (Confirmed, PR+CR) | 84.6% | 72.7% | 79.2% |
| Disease Control Rate (PR+CR+SD) | 92.3% | 100.0% | 95.8% |
| Median Progression-Free Survival (days) | Not Reached | Not Reached | Not Reached |
| Median Overall Survival (days) | Not Reached | Not Reached | Not Reached |
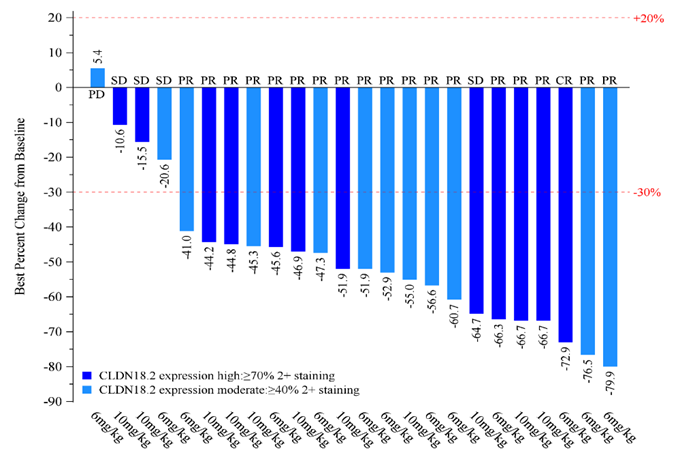
In addition to demonstrating deep and durable anti-tumor activity, ASKB589 also demonstrated good safety and tolerability. As of April 25, 2023, with a total of 106 patients enrolled in the study, escalation of monotherapy treatment has reached a dose of 20mg/kg, and escalation of ASKB589 in combination with chemotherapy has reached a dose of 15mg/kg with no dose-limiting toxicity (DLT) having been observed. Thus, a maximum tolerated dose (MTD) for ASKB589, either as monotherapy or in combination with chemotherapy, has not yet been reached. In addition, for treatment with ASKB589 at 6 and 10 mg/kg combined with CAPOX, the majority of adverse events (AEs) were grade 1-2, and the most common AEs were GI toxicities, including nausea, vomiting, loss of appetite, and hypoalbuminemia.
Dr. Lin Shen, the PI of this clinical study, commented: “This latest clinical phase II data reflects the success of ASKB589’s design as an antibody with stronger target binding activity and ADCC function than its competitors. Both the 6 mg/kg and 10 mg/kg dose groups have shown encouraging safety and efficacy. We look forward to seeing the differentiating advantages of this innovative drug against this high potential target. We hope to advance this drug to pivotal clinical trials as soon as possible, bringing hope to gastric cancer patients.”
AskGene has also developed a CLDN18.2 immunohistochemistry-based companion diagnostic kit with high sensitivity and specificity, which will be used to select patients for treatment in future clinical trials.
Conclusions
ASKB589 at a dose of ≥ 6 mg/kg combined with CAPOX has demonstrated encouraging anti-tumor activity, in addition to good safety and tolerability, for the first-line treatment of patients with advanced/metastatic G and GEJ cancer that is CLDN 18.2 positive. ASKB589 is therefore positioned to become a next-generation anti-CLDN18.2 antibody.
Gastric cancer is one of the most common cancers in the world with more than 1 million new cases and 769,000 deaths each year worldwide. It ranks fifth in incidence rate among malignant tumors and fourth in mortality. According to data from the World Health Organization, there were approximately 480,000 new gastric cancer cases and 370,000 deaths in China in 2020, accounting for 44% and 48.6% of the global gastric cancer new cases and deaths, respectively.
About ASKB589
ASKB589 is an innovative biological drug discovered and developed by AskGene. It is a recombinant humanized monoclonal antibody targeting CLDN18.2. ASKB589 is among the top 3 most advanced therapies against this target with no marketed drugs yet. ASKB589 mediates antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) through high-affinity binding to cancer cells. Enrollment in dose escalation for ASKB589 as monotherapy and in combination with CAPOX chemotherapy was completed in 2022. Enrollment continues in expansion cohorts of first-line advanced or metastatic patients with CLDN18.2-positive G&GEJ cancers that are being treated with ASKB589 in combination with chemotherapy. In addition to G&GEJ cancers, ASKB589 is being tested in clinical trials for other CLDN18.2-positive solid tumors such as pancreatic cancer. A pivotal study of ASKB589 in the treatment of gastric cancer is being planned.
About AskGene Pharma
AskGene Pharma, founded in 2012 in Los Angeles, California, is dedicated to the discovery and development of novel antibody and fusion protein therapeutics. It has established the proprietary SmartKine® cytokine prodrug platform, which significantly improves the developability of cytokines for oncology and inflammation indications. AskGene has multiple programs in clinical and preclinical developments. In addition to the first-in-class cytokine prodrug programs in clinical state, AskGene is actively advancing several programs with the potential of being best-in-class into late-stage clinical development, including ASKB589 and ASKG712, a dual-targeting antibody for the treatment of ophthalmology indications.
Forward-looking statement Disclaimer
This website contains forward-looking statements that involve risks, uncertainties and assumptions. These forward-looking statements reflect the Company’s views at the time such statements were made with respect to future events and are not a guarantee of future performance or developments. All statements other than statements of historical fact may be deemed as forward-looking, including but not limited to: any statements regarding plans, strategies and objectives for the future management of operations, including but not limited to, our clinical development and commercialization plans; predictions of any financial information; any statement of historical results that may indicate trends in our business development; statements of any expectations or beliefs regarding future events, potential markets or market size, technological developments, product lines, clinical data, results, experiments or their implications, enforceability of intellectual property rights, competitive advantage or our position in the industry; and any hypothetical statements of the items mentioned.